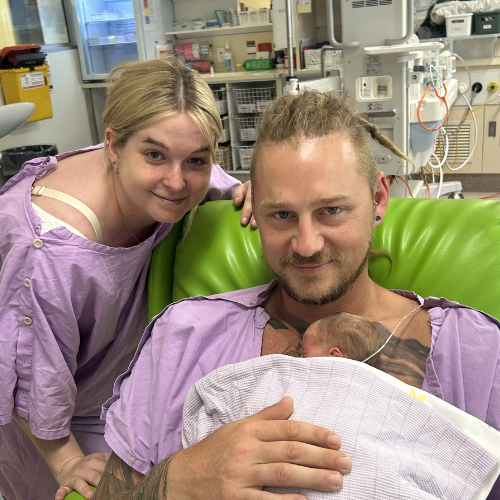
A huge thank you to leading audit and accounting firm, BDO Australia, for its extraordinary effort in raising almost $60,000 for the CAR T Cell therapy program at Royal Brisbane and Women's Hospital (RBWH).
RBWH is one of just six adult cancer centres in Australia accredited to provide the new generation cancer treatment.
RBWH Foundation CEO Simone Garske said BDO’s support would advance this innovative RBWH CAR T Cell Therapy Program to the next stage: funding the manufacture of CAR T cell treatment 'building blocks' in Brisbane, using USA/European technology quickly, cost-effectively and close to home.
“Thank you for your generous and kind support in helping us reach our goal to fund this new generation of cancer treatment,” said Ms Garske.
CAR T cell therapy research is close to the BDO team’s hearts as they honour the memory of former Client Relations Executive Director, Penny Flynn (pictured below), who bravely battled Acute Lymphoblastic Leukemia (ALL).
Despite undergoing chemotherapy and bone marrow treatment at RBWH, Penny's hopes for CAR T Cell therapy were cut short when she passed away on 11 December 2020.
BDO has set a target of $100,000 to raise funds and awareness of the importance of CAR T therapy, exemplifying the power of corporate giving and its impact on healthcare and research.



Chimeric antigen receptor (CAR) T cell therapy involves extracting a patient’s immune cells, called T cells, and genetically reprogramming them to recognise and destroy cancer cells. The reprogrammed CAR T cells are then infused back into the patient’s bloodstream.
CD19 CAR T cells have proven very effective in the treatment of B-cell blood cancers, including leukaemia and lymphoma, but there are strict criteria to access the therapy through the Medicare Benefits Schedule. Patients must have particular subtypes of B cell cancer, have tried and failed a certain number of lines of prior treatment, and fall within specific age brackets.
The process also requires the manufacture of a dedicated CAR T cell product for each patient. These publicly funded CAR T cells are manufactured commercially in the USA, which takes 4 – 8 weeks to turn around and cost about $550,000 per patient.
A new Australian-first clinical trial at RBWH, using German technology, can manufacture CAR T cells in 12 days at a cost of about $50,000 per patient.
“This lower-cost rapid turnaround in-house program enables us to provide CD19 CAR T cell therapy to patients who are ineligible for publicly funded CARs or who are too unwell to wait for CARs to be made overseas,” said RBWH Senior Staff Specialist Dr Siok Tey.
RBWH is currently the only centre in Australia with such a program and has received referrals from all around Queensland as well as interstate.
“Our vision is to make CAR T cells more effective, accessible and financially sustainable,” said Dr Tey.
“We are developing new types of CARs and we really hope that this research program can be supported because there is so much more that we want to deliver for our patients.”
CAR T cell technology is also highly effective in myeloma, but this is not currently available in Australia. Research is also underway to develop CAR T cells for common cancers such as bowel cancers, through to rarer cancers, such as brain cancers and childhood cancers,
The RBWH Foundation is honoured to support RBWH in its goal is to become a national leader in CAR T cell cancer care and research.
To learn more, or to support this new generation of cancer treatment at RBWH in Penny's honour, please click here.